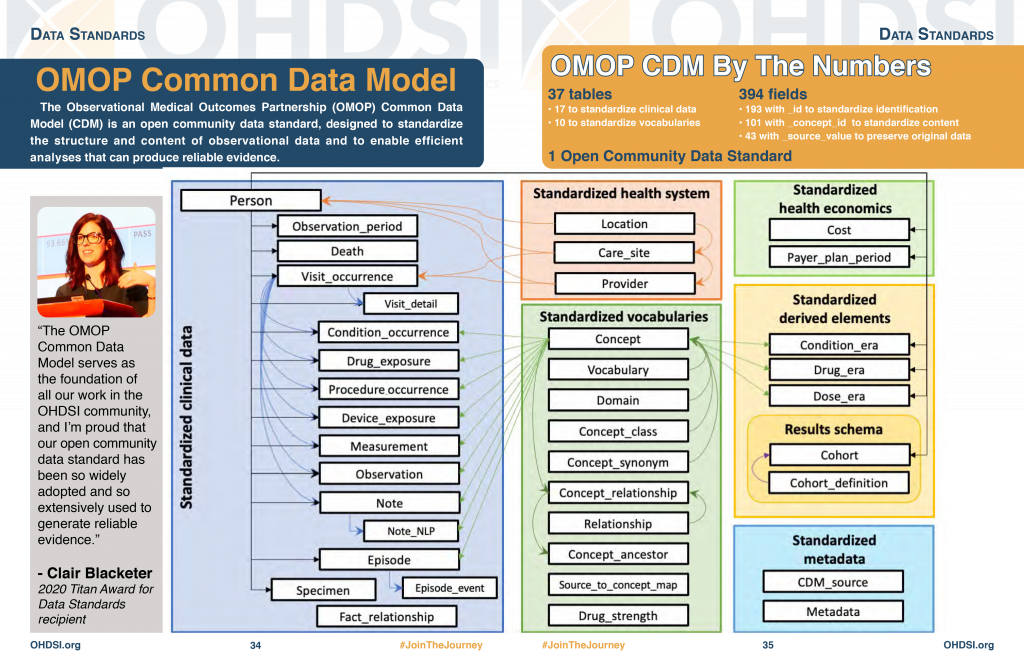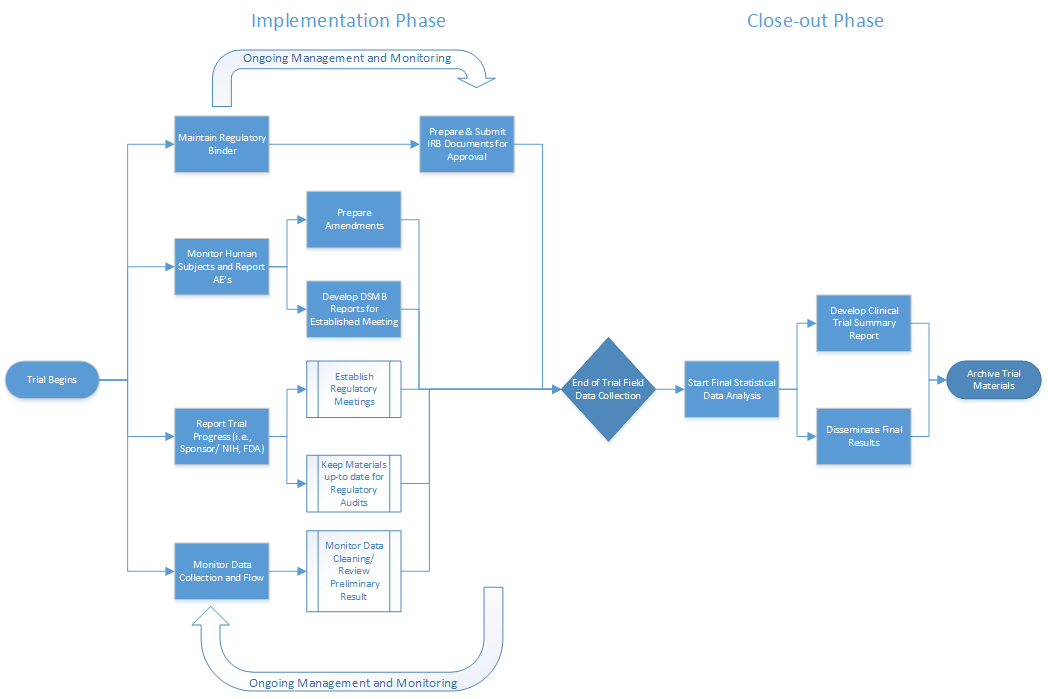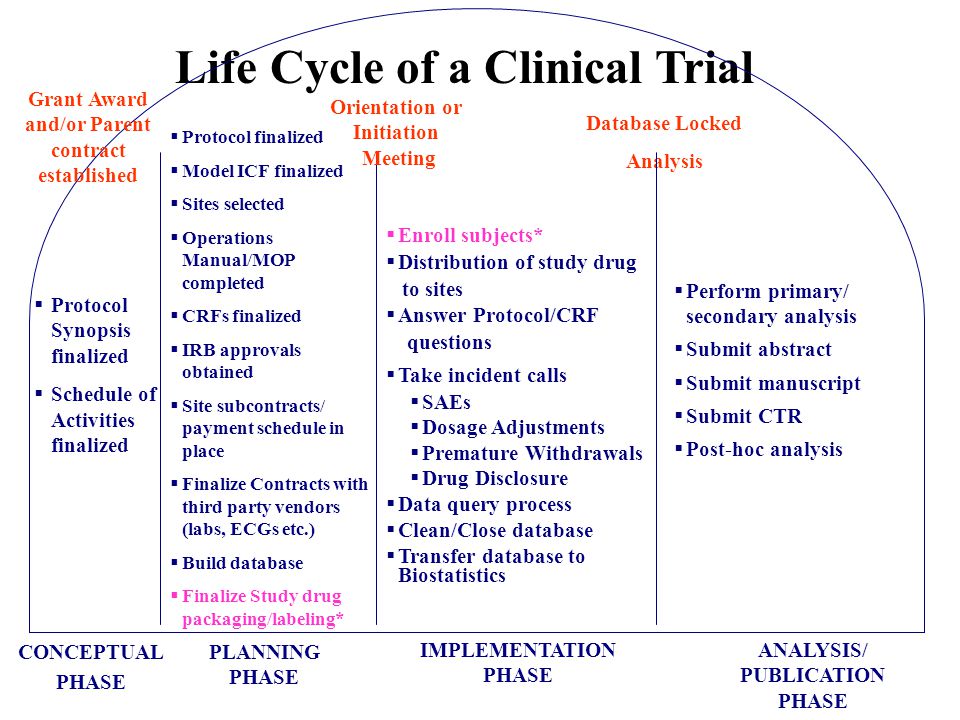
Clinical Trial Implementation Strategies: From a Coordinating Center Perspective Cornelia Kamp, MBA MSG Annual Meeting Tuesday, September 16, ppt download

PPT - A Blueprint For Clinical Research: Standard Operating Procedures PowerPoint Presentation - ID:136612

Creating an academic research organization to efficiently design, conduct, coordinate, and analyze clinical trials: The Center for Clinical Trials & Data Coordination - ScienceDirect
![PDF] Mini-implant supported canine retraction with micro-osteoperforation : A split-mouth exploratory randomized clinical trial | Semantic Scholar PDF] Mini-implant supported canine retraction with micro-osteoperforation : A split-mouth exploratory randomized clinical trial | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e6d52d250d41975e9771b89d5d2d87c8473c3853/4-Table1-1.png)
PDF] Mini-implant supported canine retraction with micro-osteoperforation : A split-mouth exploratory randomized clinical trial | Semantic Scholar

PDF) Strategies for dealing with missing data in clinical trials: from design to analysis | James Dziura and Zhixuan Fu - Academia.edu
![Prime Screen - [1 Pack] 18 Panel Urine Drug Test Cup - Instant Urine Drug Testing Marijuana (THC), AMP, BAR, BUP, BZO, COC, mAMP, MDMA, MOP, MTD, OXY, PCP, TCA, EtG, FTY, Prime Screen - [1 Pack] 18 Panel Urine Drug Test Cup - Instant Urine Drug Testing Marijuana (THC), AMP, BAR, BUP, BZO, COC, mAMP, MDMA, MOP, MTD, OXY, PCP, TCA, EtG, FTY,](https://i5.walmartimages.com/seo/Prime-Screen-1-Pack-18-Panel-Urine-Drug-Test-Cup-Instant-Testing-Marijuana-THC-AMP-BAR-BUP-BZO-COC-mAMP-MDMA-MOP-MTD-OXY-PCP-TCA-EtG-FTY-TRA-K2-KRA-T_4f53f71f-e904-4750-bb78-dd75372d37fe.31921e6fdebba9464e479eceb3265143.jpeg?odnHeight=768&odnWidth=768&odnBg=FFFFFF)