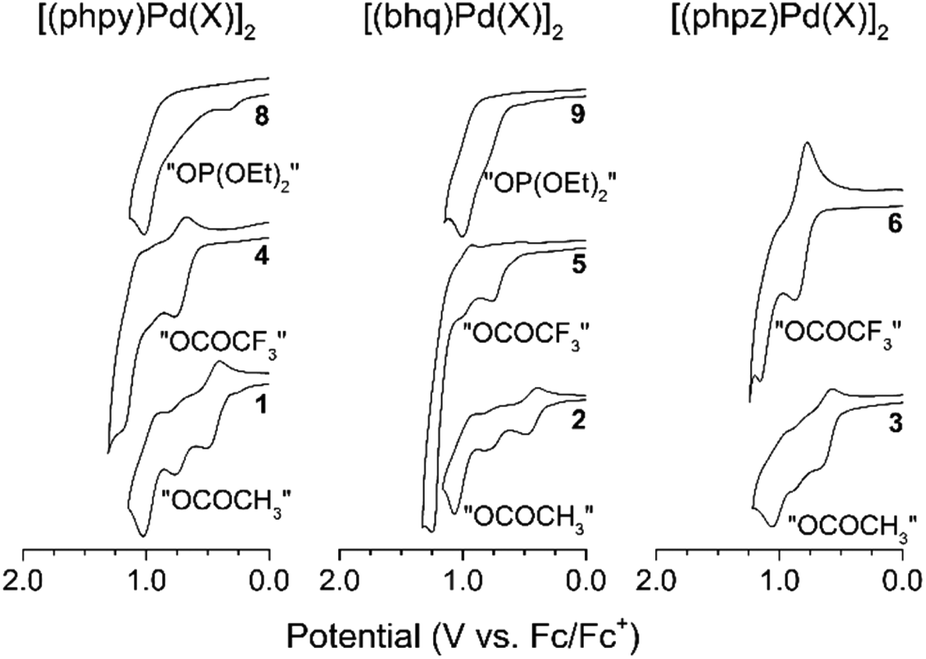
Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society
CV and redox potential for the Fe(III)/Fe(II) redox couple of different... | Download Scientific Diagram

How palladium inhibits CO poisoning during electrocatalytic formic acid oxidation and carbon dioxide reduction | Nature Communications

Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K

Polyhedron shaped palladium nanostructures embedded on MoO2/PANI-g-C3N4 as high performance and durable electrocatalyst for oxygen reduction reaction - ScienceDirect

Effect of palladium and its nanogeometry on the redox electrochemistry of tetracyanoquinodimethane modified electrode; application in electrochemical sensing of ascorbic acid | Journal of Applied Electrochemistry

Revealing the Influence of Diverse Secondary Metal Cations on Redox‐Active Palladium Complexes - Golwankar - 2022 - Chemistry – A European Journal - Wiley Online Library

Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society

A Distinctive Pattern for Substituent Effects on Transition Metal Centers: Enhanced Electron-Donating Capacity of Cationic Palladium Species | CCS Chemistry

In-situ oxidation of Palladium–Iridium nanoalloy anchored on Nitrogen-doped graphene as an efficient catalyst for methanol electrooxidation - ScienceDirect

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society

Chemically activating MoS2 via spontaneous atomic palladium interfacial doping towards efficient hydrogen evolution | Nature Communications

Hydrometallurgical recycling of palladium and platinum from exhausted diesel oxidation catalysts - ScienceDirect

Electrochemical Methods for Pd‐catalyzed C−H Functionalization - Erchinger - 2021 - Asian Journal of Organic Chemistry - Wiley Online Library

Palladium-Catalyzed Reductive Homocoupling of Aromatic Halides and Oxidation of Alcohols | The Journal of Organic Chemistry






