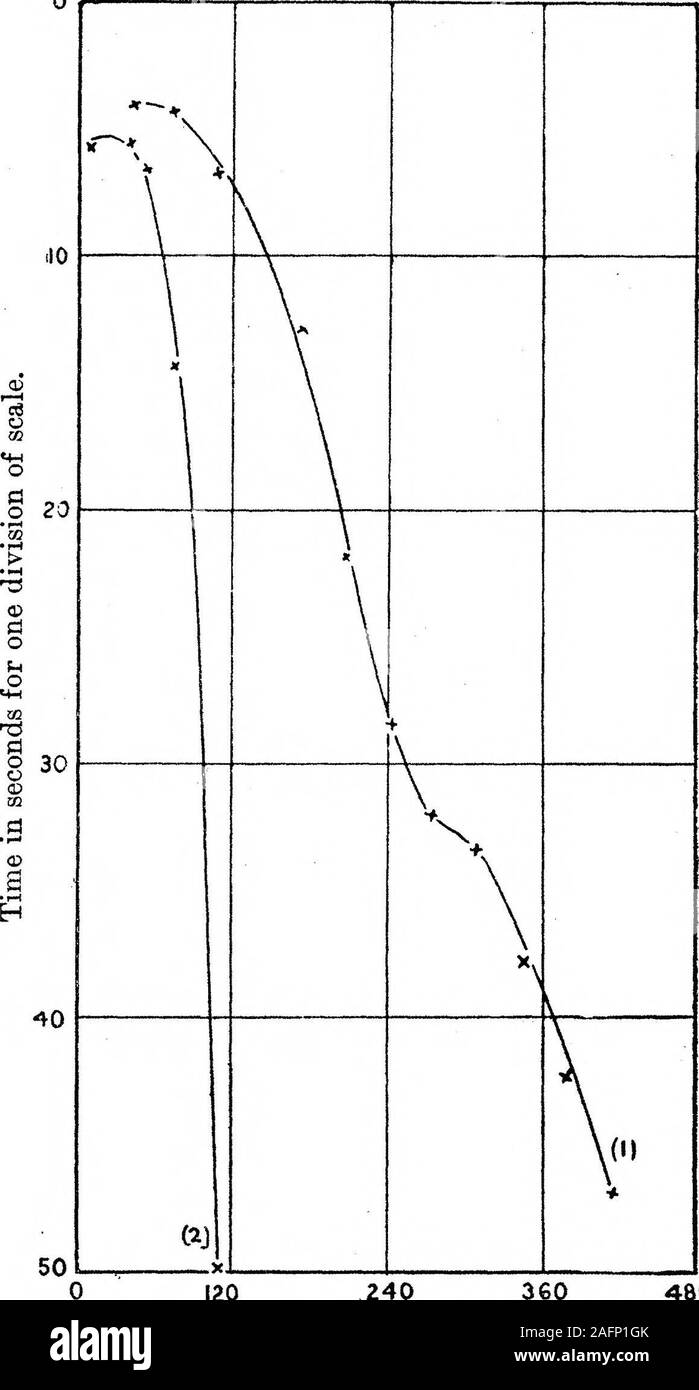
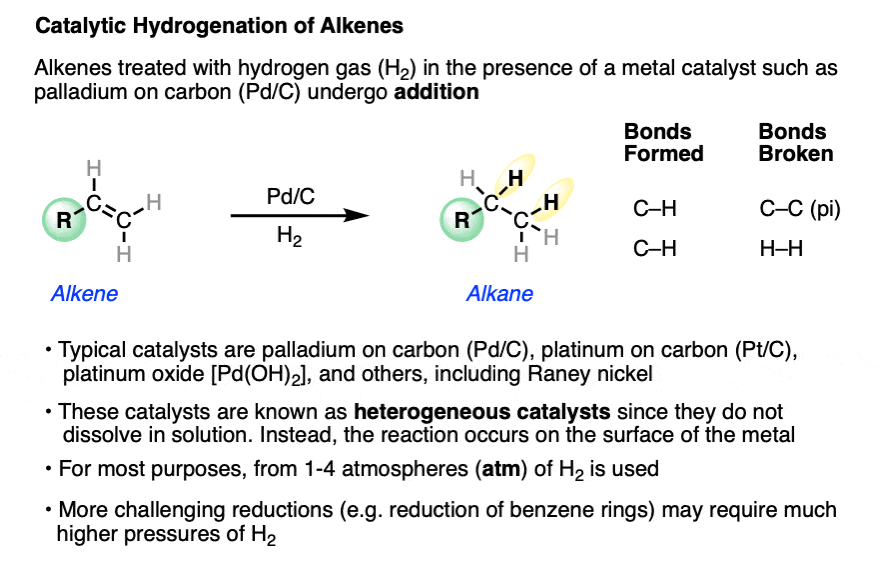
Sub-second and ppm-level optical sensing of hydrogen using templated control of nano-hydride geometry and composition | Nature Communications

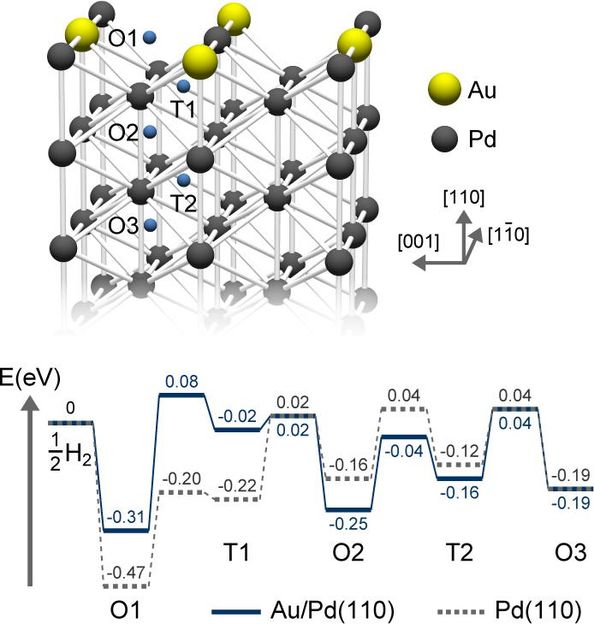
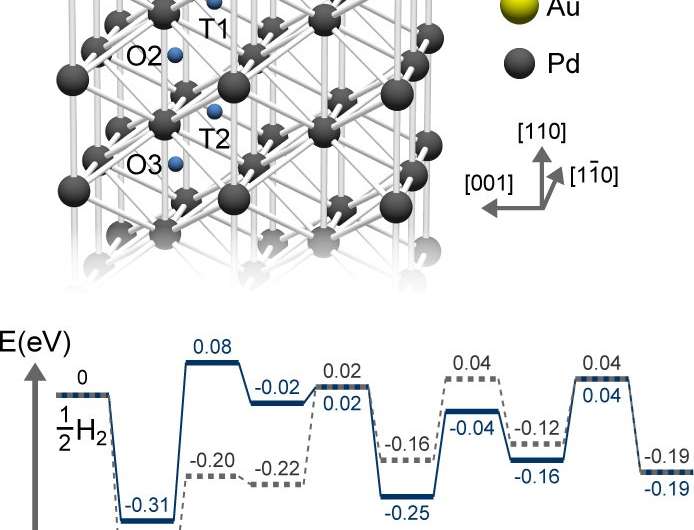
DFT Study of Hydrogen Adsorption on Palladium Decorated Graphene | The Journal of Physical Chemistry C

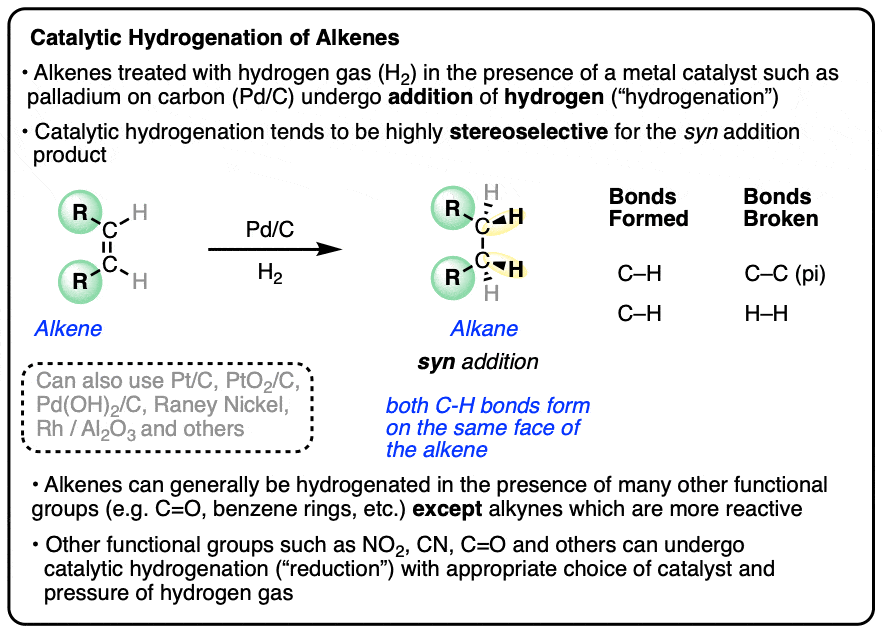
Hydrogen in Palladium and Storage Properties of Related Nanomaterials: Size, Shape, Alloying, and Metal‐Organic Framework Coating Effects - Dekura - 2019 - ChemPhysChem - Wiley Online Library

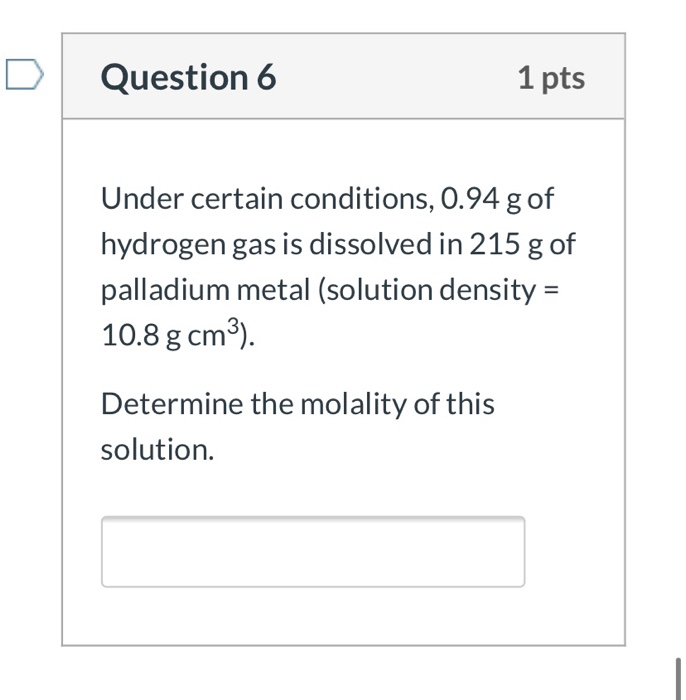
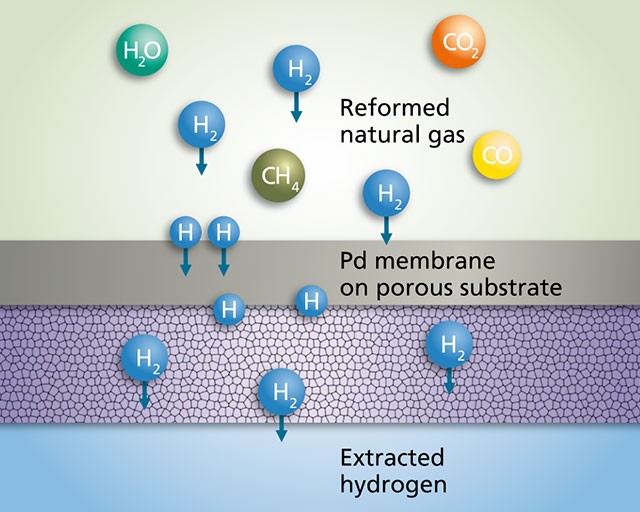
Solid palladium metal is known to actually absorb hydrogen gas, and no other. This behavior is used to make ultra, ultra-pure hydrogen. (In fact, because of this, hydrogen can be obtained in