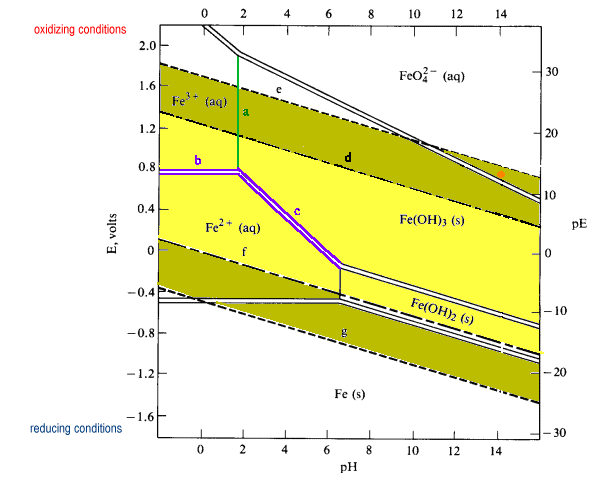
Eh-pH Diagram showing palladium and iron species and water stability... | Download Scientific Diagram
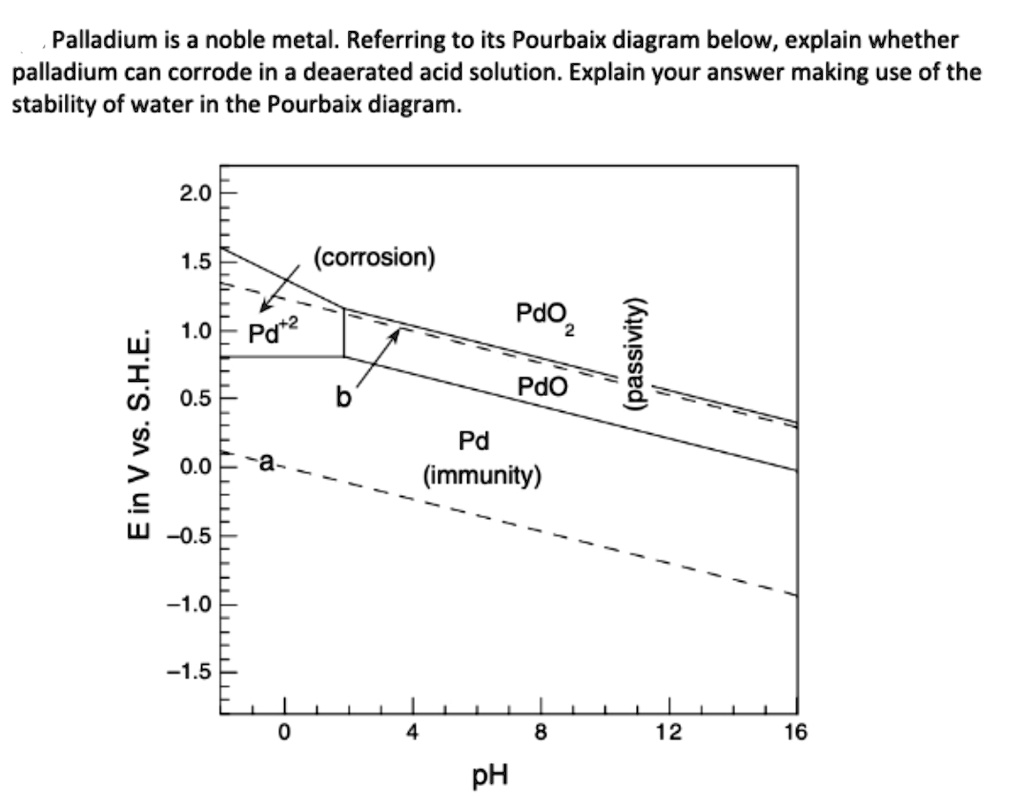
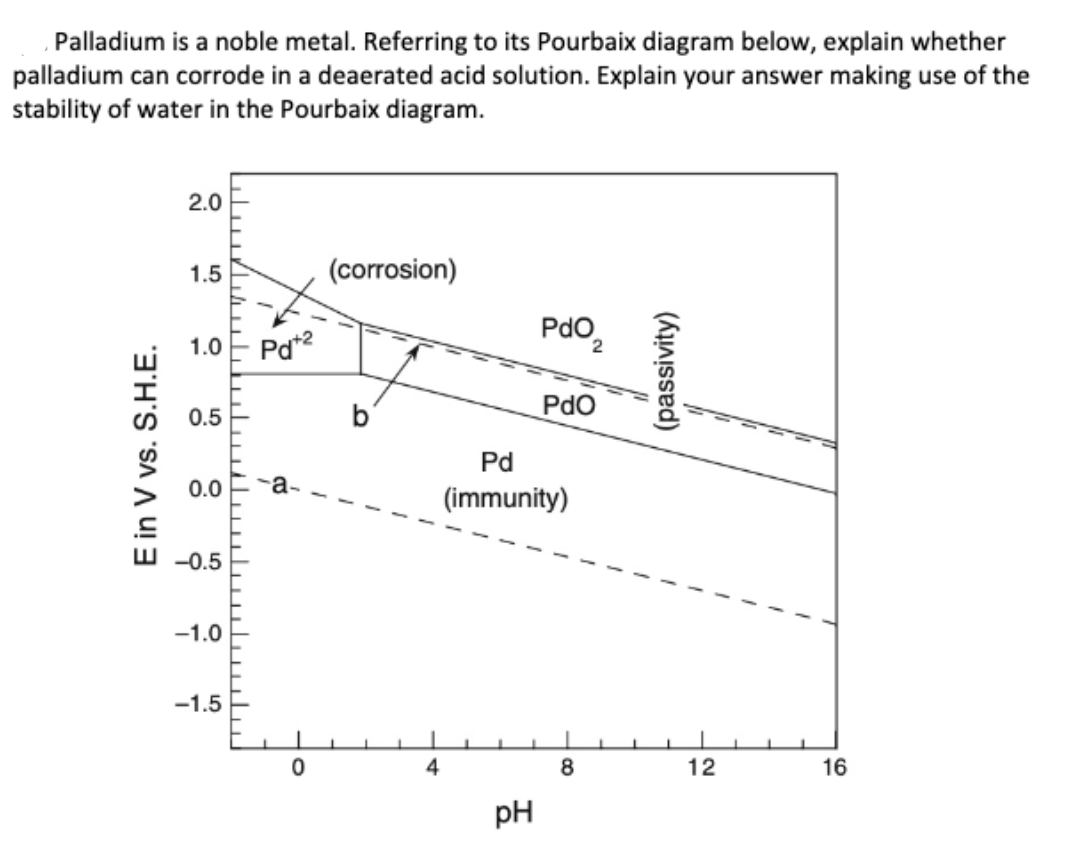
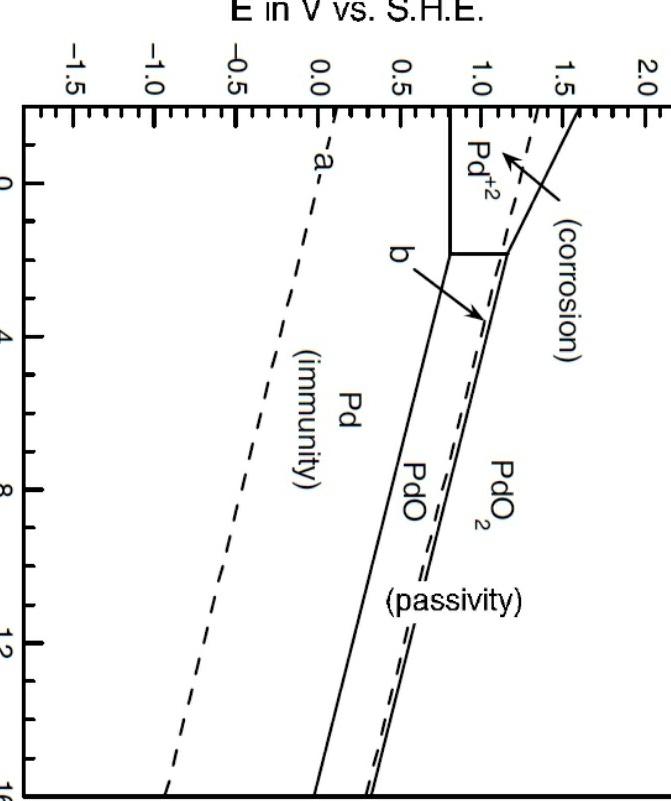
![Pourbaix diagram for palladium at a metal ion concentration of 10-5 M [4]. | Download Scientific Diagram Pourbaix diagram for palladium at a metal ion concentration of 10-5 M [4]. | Download Scientific Diagram](https://www.researchgate.net/profile/Stuart-Lyon-3/publication/286129486/figure/fig3/AS:635155370758144@1528444249778/Pourbaix-diagram-for-platinum-at-a-metal-ion-concentration-of-10-5-M-4_Q320.jpg)
Pourbaix diagram for palladium at a metal ion concentration of 10-5 M [4]. | Download Scientific Diagram
The dissolution of palladium as a function of glucose concentration in chloride containing solutions of acidic pH
![Pourbaix diagram for palladium at a metal ion concentration of 10-5 M [4]. | Download Scientific Diagram Pourbaix diagram for palladium at a metal ion concentration of 10-5 M [4]. | Download Scientific Diagram](https://www.researchgate.net/profile/Stuart-Lyon-3/publication/286129486/figure/fig4/AS:635155370737669@1528444249855/Pourbaix-diagram-for-palladium-at-a-metal-ion-concentration-of-10-5-M-4_Q320.jpg)
Pourbaix diagram for palladium at a metal ion concentration of 10-5 M [4]. | Download Scientific Diagram
Ordered Intermetallic Nanoparticles with High Catalytic Activity Prepared by an Electrochemically Induced Phase Transformation

Complexation of platinum, palladium and rhodium with inorganic ligands in the environment | Geochemistry: Exploration, Environment, Analysis
The dissolution of palladium as a function of glucose concentration in chloride containing solutions of acidic pH

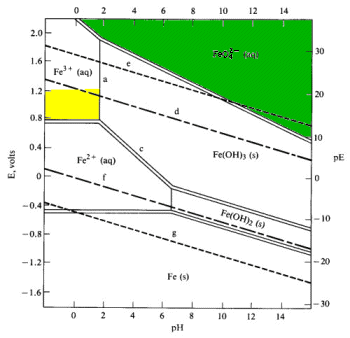
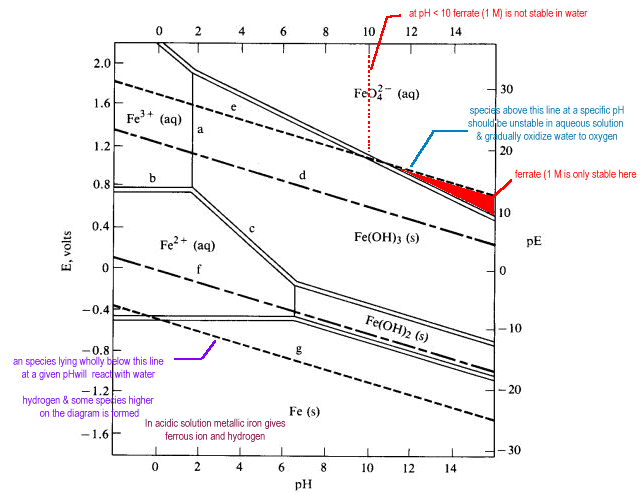
Pourbaix diagram showing iron and palladium species and water stability... | Download Scientific Diagram

Quantifying Confidence in DFT-Predicted Surface Pourbaix Diagrams of Transition-Metal Electrode–Electrolyte Interfaces | Langmuir

Pourbaix diagram showing iron and palladium species and water stability... | Download Scientific Diagram

Electrochemical nucleation of palladium on graphene: A kinetic study with an emphasis on hydrogen co-reduction - ScienceDirect
![Pourbaix diagram for palladium at a metal ion concentration of 10-5 M [4]. | Download Scientific Diagram Pourbaix diagram for palladium at a metal ion concentration of 10-5 M [4]. | Download Scientific Diagram](https://www.researchgate.net/publication/286129486/figure/fig4/AS:635155370737669@1528444249855/Pourbaix-diagram-for-palladium-at-a-metal-ion-concentration-of-10-5-M-4.png)
Pourbaix diagram for palladium at a metal ion concentration of 10-5 M [4]. | Download Scientific Diagram