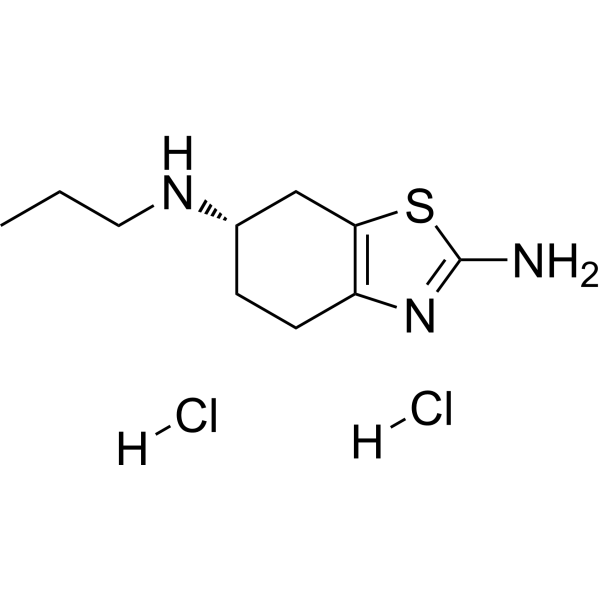
Pramipexole for the treatment of depressive symptoms in patients with Parkinson's disease: a randomised, double-blind, placebo-controlled trial - The Lancet Neurology
SÚHRN CHARAKTERISTICKÝCH VLASTNOSTÍ LIEKU 1. NÁZOV LIEKU Pramipexol Teva Pharma 0,26 mg Pramipexol Teva Pharma 0,52 mg Pram
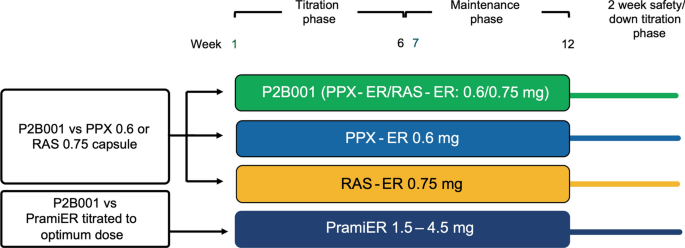
P2B001 (Extended Release Pramipexole and Rasagiline): A New Treatment Option in Development for Parkinson's Disease | Advances in Therapy
Písomná informácia pre používateľa Pramipexol Teva Pharma 0,26 mg Pramipexol Teva Pharma 0,52 mg Pramipexol Teva Pharma 1,